Optimizing a PK assay for reliable data in clinical studies
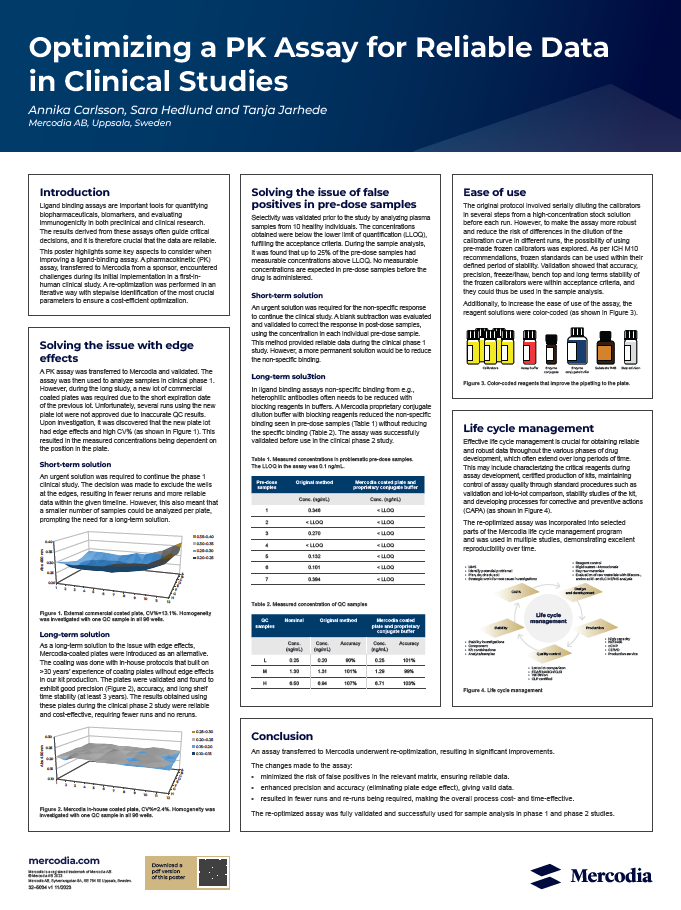
This poster presents the successful re-optimization of a PK assay transferred to Mercodia from a sponsor which initially experienced issues during a first-in-human clinical study. The goal of the re-optimization process was to overcome these challenges and ensure the generation of reliable data while maintaining cost-efficiency.
The improvements made to the assay:
- Minimized the risk of false positives in the relevant matrix, ensuring reliable data
- Enhanced precision and accuracy (eliminating plate edge effect), giving valid data
- Resulted in fewer runs and re-runs being required, making the overall process cost- and time-effective
The re-optimized PK assay was rigorously validated and successfully utilized for sample analysis in both phase 1 and phase 2 clinical studies.
Download poster
Ligand binding assays are important tools for quantifying biopharmaceuticals and biomarkers, and evaluating immunogenicity in both preclinical and clinical research. The results derived from these assays often guide critical decisions, and it is therefore crucial that the data are reliable.
This poster highlights some key aspects to consider when improving a ligand-binding assay. A pharmacokinetic (PK) assay, transferred to Mercodia from a sponsor, encountered challenges during its initial implementation in a first-in-human clinical study. A re-optimization was performed in an iterative way with stepwise identification of the most crucial parameters to ensure a cost-efficient optimization.
Related material
Are you involved in drug development or bioanalysis? Don’t miss our webinar, hosted by AAPS, where we delve into the crucial aspects of developing a robust ligand binding assay.
Watch this free webinar where we show real-life common challenges and suggest solutions based on our extensive experience in developing ligand binding assays.
Watch webinar: Risk Mitigation in Bioanalysis to Maintain the Integrity of Drug Development Studies
CONTACT US

Customer Success Manager & Area Sales Manager Nordics
robert.almstedt@mercodia.com (+46) 72 292 15 08